(C) 2012 Mohammed A. Hannan. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
The large carpenter bees (Xylocopinae, Xylocopa Latreille) occurring in central Saudi Arabia are reviewed. Two species are recognized in the fauna, Xylocopa (Koptortosoma) aestuans (Linnaeus) and Xylocopa (Ctenoxylocopa) sulcatipes Maa. Diagnoses for and keys to the species of these prominent components of the central Saudi Arabian bee fauna are provided to aid their identification by pollination researchers active in the region. Females and males of both species are figured and biological notes provided for Xylocopa sulcatipes. Notes on the nesting biology and ecology of Xylocopa sulcatipes are appended. As in studies for this species from elsewhere, nests were found in dried stems of Calotropis procera (Aiton) (Asclepiadaceae) and Phoenix dactylifera L. (Arecaceae).
Apoidea, Anthophila, Xylocopini, Arabian Peninsula, systematics, biology, host plants, nesting
The tribe Xylocopini comprises the large carpenter bees (Xylocopinae: Xylocopa Latreille) species of which principally nest in dead wood (including the wood of human constructions), bamboo culms, and other similar substrates (e.g., Hurd 1958, 1978; Hurd and Moure 1960, 1963; Sakagami and Laroca 1971; Gerling et al. 1983, 1989; Maeta et al. 1985, 1996; Raju and Rao 2006; Boontop et al. 2008; Gonzalez et al. 2009; Pereira and Garófalo 2010; Prager and Hunter 2011), and Le Goff (2004) even records a species emerging from the wood of a Senegalese ceremonial mask. Owing to their conspicuousness, interesting biology and behavior, and agricultural potential (e.g., Keasar 2010), the group has received increased systematic scrutiny, although most recent work has focused on relationships among the tribes of the subfamily or those of the constituent genera and subgenera (e.g., Minckley 1998; Engel 2001, in press; Leys et al. 2002; Flores-Prado et al. 2010). Meanwhile, the number of detailed, species-level revisions has lagged over the last 15 years (although when done, there are some very nice examples: e.g., Leys 2000) despite the fact that it is at this level for which the documentation and interpretation of biological phenomena is most critical (e.g., Engel 2011). Today, the diversity of carpenter bees in many areas of the world remains largely unexplored.
As part of an on-going effort to survey the bee fauna and pollinator resources of the Kingdom of Saudi Arabia and to eliminate the taxonomic impediment for working on this diverse region, we have begun with surveys of the melittofauna from central Saudi Arabia. Herein we provide a brief contribution to this larger effort by documenting the species of the large carpenter bees occurring in this area and as an aid to studies of wild bee pollination already underway (Hannan et al. in prep.). Two species are recognized from the region, Xylocopa (Koptortosoma) aestuans (Linnaeus) and Xylocopa (Ctenoxylocopa) sulcatipes Maa, although the latter may be frequently found misidentified in some collections as Xylocopa (Xylomelissa) hottentotta Smith or XC.) fenestrata (Fabricius) (e.g., Al-Ahmadi and Salem 1999). Xylocopa hottentotta is an entirely unrelated species of African distribution (Eardley 1983, 1993), while Xylocopa fenestrata is certainly very closely allied to Xylocopa sulcatipes but occurs more easterly and southerly (Maa 1970). The latter species is quite similar but can be readily distinguished on the basis of the male terminalia (Maa 1970, and figures herein, vide infra). In total five nominal species have been recorded from throughout the Arabian Peninsula [e.g., Al-Ahmadi and Salem 1999: recorded as Xylocopa aestuans, Xylocopa hottentotta, Xylocopa caffra (Linnaeus), and Xylocopa valga?], but many of these seem to be misidentifications (e.g., Xylocopa hottentotta) and thorough collecting and new identifications, particularly with comparisons to holotypes, is desperately needed. Diagnoses, figures (particularly the male terminalia), and keys are provided so as to aid regional entomologists in the identification of their material. In addition, we append observations on the biology and ecology of Xylocopa sulcatipes.
Material and methodsMaterial examined herein is deposited in the King Saud University Museum of Arthropods, Plant Protection Department, College of Food and Agriculture Sciences, King Saud University, Riyadh, Kingdom of Saudi Arabia (KSMA) and Division of Entomology (Snow Entomological Collections), University of Kansas Natural History Museum, Lawrence, Kansas, USA (SEMC). Photomicrographs were prepared using a Nikon D1x digital camera attached to an Infinity K-2 long-distance microscope lens. Morphological terminology in the diagnoses follows that of Engel (2001) and Michener (2007). Herein we follow the supraspecific classification of Xylocopini advocated by Minckley (1998) and Michener (2007).
The nesting biology of Xylocopa sulcatipes was studied in Amariah, approximately 25 km northwest of Riyadh, from September 2010 through December 2011. Nests were found on 6 June 2011 at the base of a large hill near an agricultural farm near Wadi Amariah and the highway to Riyadh. Prior to collection the nests were observed for at least an hour to note the coming and going of bees. Most nests were located around 9:00am and collected around 12:00pm. Nests were sealed with plastic and brought to the lab for dissection and study. During four visits (6, 12, 19 June and 28 September 2011) a total of 13 nests were collected (Table 1). Nests were in the dead branches of local milkweeds [Calotropis procera (Aiton) (Asclepiadaceae), more widely known as the “Apple of Sodom”] growing in a sparsely vegetative desert area and among date palms, Phoenix dactylifera L. (Arecaceae). Nests were measured, sketched, and photographed, and the inhabitants deposited in the KSMA repository.
Measurements of sampled nests of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia collected in dead wood of two plants. Means are given with standard deviations. n = number of nests sampled for each metric.
| Asclepiadaceae | Arecaceae | |
|---|---|---|
| Metric | Calotropis procera (Aiton) | Phoenix dactylifera L. |
| Branch length (cm) | 147.75±74.59 (n=8) | 57.33±6.53 (n=6) |
| Nest entrance (mm) | 9.06±0.78 X 8.85±0.86 (n=8) | 10.17±2.17 X 11±2 (n=7) |
| Height of nest from ground (cm) | 83.50±30.3 (n=8) | 400±0 (n=6) |
| Length of nest (cm) | 23.61±14.93 (n=7) | 11.05±4.36 (n=6) |
| Branch diameter at nest (cm) | 1.88±0.38 (n=8) | 6.83±0.41 (n=6) |
| Internal diameter of nest (cm) | 1.28±0.19 (n=8) | 1.77±0.12 (n=6) |
| Number of cells/nest | 6.60±5.6 (n=5) | 4.50±1.64 (n=6) |
| Length of cells (mm) | 18.8±1.63 (n=30) | 20.11±1.37 (n=19) |
| Asclepiadaceae | Arecaceae | |
|---|---|---|
| Metric | Calotropis procera (Aiton) | Phoenix dactylifera L. |
| Branch length (cm) | 147.75±74.59 (n=8) | 57.33±6.53 (n=6) |
| Nest entrance (mm) | 9.06±0.78 X 8.85±0.86 (n=8) | 10.17±2.17 X 11±2 (n=7) |
| Height of nest from ground (cm) | 83.50±30.3 (n=8) | 400±0 (n=6) |
| Length of nest (cm) | 23.61±14.93 (n=7) | 11.05±4.36 (n=6) |
| Branch diameter at nest (cm) | 1.88±0.38 (n=8) | 6.83±0.41 (n=6) |
| Internal diameter of nest (cm) | 1.28±0.19 (n=8) | 1.77±0.12 (n=6) |
| Number of cells/nest | 6.60±5.6 (n=5) | 4.50±1.64 (n=6) |
| Length of cells (mm) | 18.8±1.63 (n=30) | 20.11±1.37 (n=19) |
This is the largest and most widespread subgenus of carpenter bees, with at least 196 recognized species ranging throughout Subsaharan Africa to the Mediterranean countries of that continent, Dalmatia, the Arabian Peninsula, southwestern Asia, and southern Asia east to the Philippines, Taiwan, and Japan, and south through Indonesia, New Guinea, and the Bismarck Archipelago to southernmost Australia (Michener 2007). The subgenus can be recognized by the female mesoscutellum having a sharp truncation overhanging the metanotum (as in subgenus Mesotrichia Westwood) and surpassing the posterior margin of the latter, and males with unmodified tegulae (elongate in Mesotrichia) (Michener 2007).
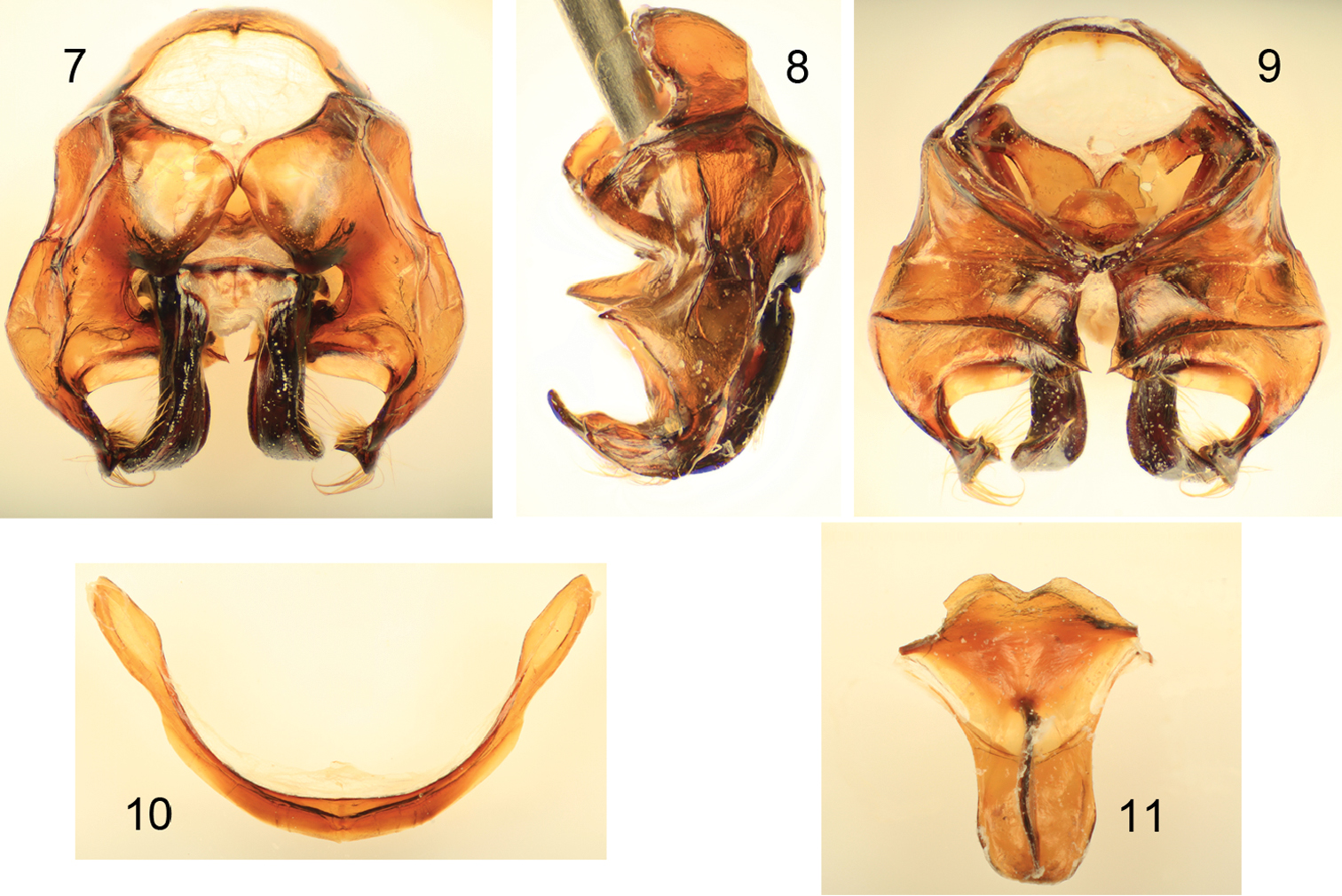
Diagnosis. Xylocopa aestuans can be most readily distinguished from other Saudi Arabian large carpenter bees by the following: female face with largely white or pale pubescence (Fig. 5), mesosomal dorsum densely covered by yellow pubescence obscuring underlying integument (Figs 1, 2); mandible bidentate at apex; posterodorsal margin of mesoscutellum projecting beyond posterior margin of metanotum; pygidial plate unarmed. Male covered by dense yellow pubescence (Figs 3, 4, 6); first metasomal tergum with subhorizontal dorsal surface abruptly and angulately separated from declivitous anterior surface; gradulus of first metasomal tergum transverse, lateral extremities not directed posteriorly; male terminalia as in figures 7–11.
Comments. Xylocopa aestuans is one of the widespread and ubiquitous of large carpenter bee species. There has been considerable debate regarding the identity of the species of Koptortosoma similar to Xylocopa aestuans (i.e., considering them synonyms, subspecies, or separate species), with different authors of varying opinions how to segregate the minor variation into natural taxonomic entities (e.g., Lieftinck 1964). The Saudi Arabian populations have been at times considered to the belong to the largely African, Xylocopa pubescens Spinola, although the genitalia of those populations are quite dissimilar from true Xylocopa pubescens. Indeed, the genitalia (Figs. 7–11) and other characters are certainly more alike the more easterly populations of Xylocopa aestuans and there seems little reason at this time to not consider the central Saudi Arabian populations as such, as was done by Shalaby (1961). The species has also been recorded from the United Arab Emirates (Harten 2005; Dathe 2009). Biological accounts, largely from India or Southeast Asia, have been provided by Dover (1924), Monod (1977), Binti (1992), El-Borollosy and Ismail (1972: note that these observations may be of Xylocopa pubescens, the identity of their material requires checking), and Punekar et al. (2010).
Habitus photomicrographs of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 1 Female, dorsal 2 Female, lateral 3 Male, dorsal 4 Male, lateral.
Habitus photomicrographs of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 1 Female, dorsal 2 Female, lateral 3 Male, dorsal 4 Male, lateral.
Faces of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 5 Female 6 Male.
Faces of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 5 Female 6 Male.
Male terminalia of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 7 Genital capsule, dorsal aspect 8 Genital capsule, lateral aspect 9 Genital capsule, ventral aspect 10 Seventh metasomal sternum 11 Eighth metasomal sternum.
Male terminalia of Xylocopa (Koptortosoma) aestuans (Linnaeus) from central Saudi Arabia. 7 Genital capsule, dorsal aspect 8 Genital capsule, lateral aspect 9 Genital capsule, ventral aspect 10 Seventh metasomal sternum 11 Eighth metasomal sternum.
This is a widespread, albeit not very diverse, subgenus of Old World carpenter bees (Maa 1970; Michener 2007). The lineage can be recognized by the prolonged posterior pronotal lobes and elevated process of the spiracles on the third metasomal tergum in males, while females are noteworthy for the combination of a row of tubercles along each margin of the metabasitibial plate, the tridentate mandibles, and a single spine on the outer apex of the metatibia.
http://species-id.net/wiki/Xylocopa_sulcatipes
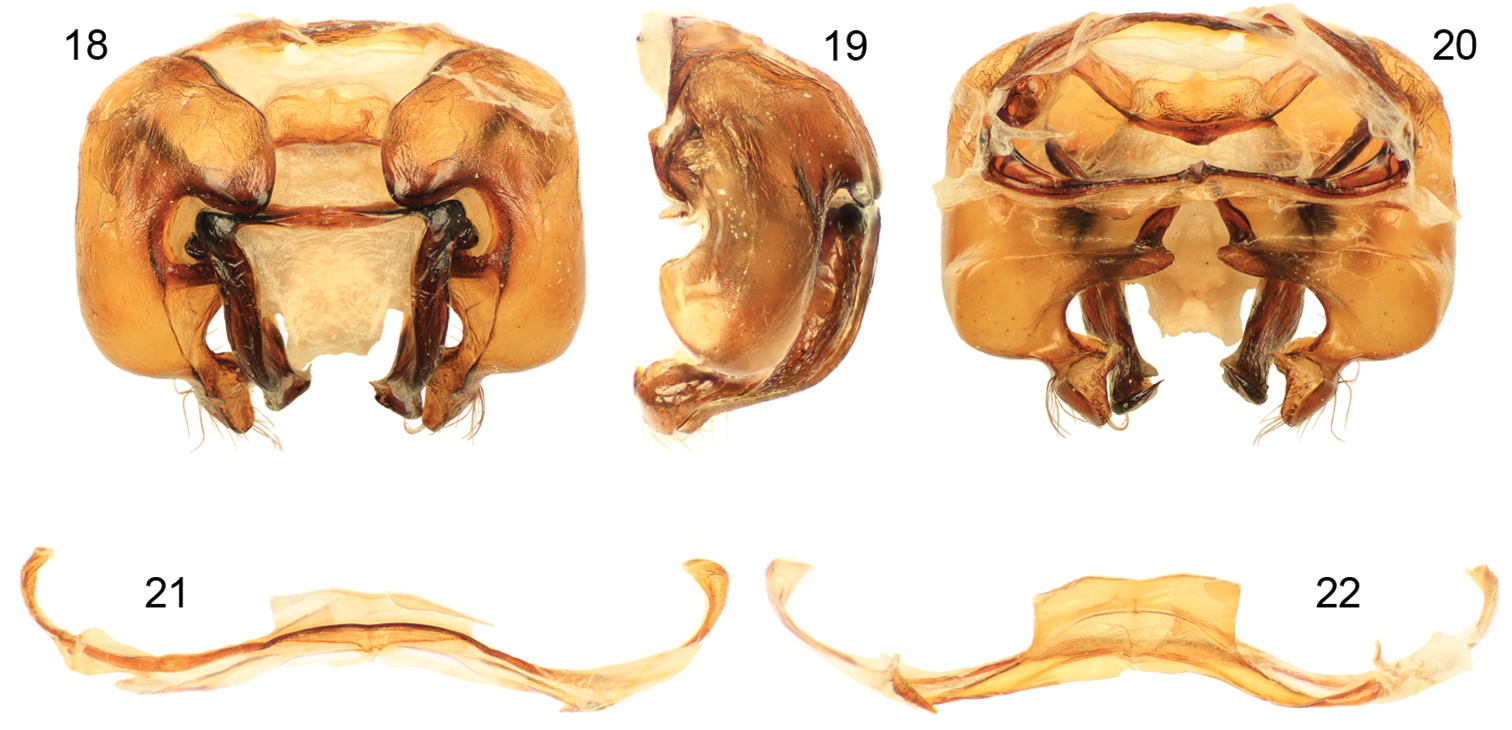
Figs 12–22Xylocopa sulcatipes can be most readily distinguished from other Arabian large carpenter bees by the following: Female with face with largely black pubescence (Fig. 16), mesosomal dorsum largely covered by black pubescence not obscuring underlying integument (Figs 12, 13); mandible tridentate at apex; mesoscutellum not projecting over metanotum, apical margin rounded in profile; pygidial plate armed on each side with subapical spine. Male covered by largely fuscous to black pubescence except face, dorsum of mesosoma, and apicolateral patches of first metasomal tergum with predominantly white or pale setae (Figs 14, 15, 17); first metasomal tergum with subhorizontal dorsal surface rounding into declivitous anterior surface; gradulus of first metasomal tergum laterally curved posteriorly; male terminalia as in figures 18–22.
Maa (1970) recorded Xylocopa sulcatipes from Saudi Arabia, Yemen, Israel, and Transcaspia (likely northern Iran, or southwesternmost Turkmenistan), while Vicidomini (2004) gave localities in Jordan. The records of Xylocopa fenestrata from the United Arab Emirates (Dathe 2009) are likely Xylocopa sulcatipes, and this material should be dissected and compared with the images herein (Figs. 18–22) as well as those of Maa (1970). We have found that Maa’s (1970) characterization of the terminalic differences holds well for observed populations and the species he recognized appear to be good (Engel pers. obs.).
Habitus photomicrographs of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 12 Female, dorsal 13 Female, lateral 14 Male, dorsal 15 Male, lateral.
Habitus photomicrographs of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 12 Female, dorsal 13 Female, lateral 14 Male, dorsal 15 Male, lateral.
Faces of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 16 Female 17 Male.
Faces of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 16 Female 17 Male.
Male terminalia of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 18 Genital capsule, dorsal aspect 19 Genital capsule, lateral aspect 20 Genital capsule, ventral aspect 21 Seventh metasomal sternum 22 Eighth metasomal sternum.
Male terminalia of Xylocopa (Ctenoxylocopa) sulcatipes Maa from central Saudi Arabia. 18 Genital capsule, dorsal aspect 19 Genital capsule, lateral aspect 20 Genital capsule, ventral aspect 21 Seventh metasomal sternum 22 Eighth metasomal sternum.
| 1 | Males | 2 |
| – | Females | 3 |
| 2 | Bee covered by dense yellow pubescence; first metasomal tergum with subhorizontal dorsal surface abruptly and angulately separated from declivitous anterior surface; gradulus of first metasomal tergum transverse, lateral extremities of gradulus not directed posteriorly; terminalia as in figures 7–11 | Xylocopa aestuans (Linnaeus) |
| – | Bee covered by largely fuscous to black pubescence except face, dorsum of mesosoma, and apicolateral patches of first metasomal tergum with predominantly white or pale setae; first metasomal tergum with subhorizontal dorsal surface rounding into declivitous anterior surface; gradulus of first metasomal tergum laterally curved posteriorly; terminalia as in figures 18–22 | Xylocopa sulcatipes Maa |
| 3 | Mesosomal dorsum densely covered by yellow pubescence obscuring underlying integument; face with largely white or pale pubescence; pygidial plate unarmed; posterodorsal margin of mesoscutellum projecting beyond posterior margin of metanotum; mandible bidentate at apex | Xylocopa aestuans (Linnaeus) |
| – | Mesosomal dorsum largely covered by black pubescence not obscuring underlying integument; face with largely black pubescence; pygidial plate armed on each side with subapical spine; mesoscutellum not projecting over metanotum, apical margin rounded in profile; mandible tridentate at apex | Xylocopa sulcatipes Maa |
| 1 | Males | 2 |
| – | Females | 3 |
| 2 | Bee covered by dense yellow pubescence; first metasomal tergum with subhorizontal dorsal surface abruptly and angulately separated from declivitous anterior surface; gradulus of first metasomal tergum transverse, lateral extremities of gradulus not directed posteriorly; terminalia as in figures 7–11 | Xylocopa aestuans (Linnaeus) |
| – | Bee covered by largely fuscous to black pubescence except face, dorsum of mesosoma, and apicolateral patches of first metasomal tergum with predominantly white or pale setae; first metasomal tergum with subhorizontal dorsal surface rounding into declivitous anterior surface; gradulus of first metasomal tergum laterally curved posteriorly; terminalia as in figures 18–22 | Xylocopa sulcatipes Maa |
| 3 | Mesosomal dorsum densely covered by yellow pubescence obscuring underlying integument; face with largely white or pale pubescence; pygidial plate unarmed; posterodorsal margin of mesoscutellum projecting beyond posterior margin of metanotum; mandible bidentate at apex | Xylocopa aestuans (Linnaeus) |
| – | Mesosomal dorsum largely covered by black pubescence not obscuring underlying integument; face with largely black pubescence; pygidial plate armed on each side with subapical spine; mesoscutellum not projecting over metanotum, apical margin rounded in profile; mandible tridentate at apex | Xylocopa sulcatipes Maa |
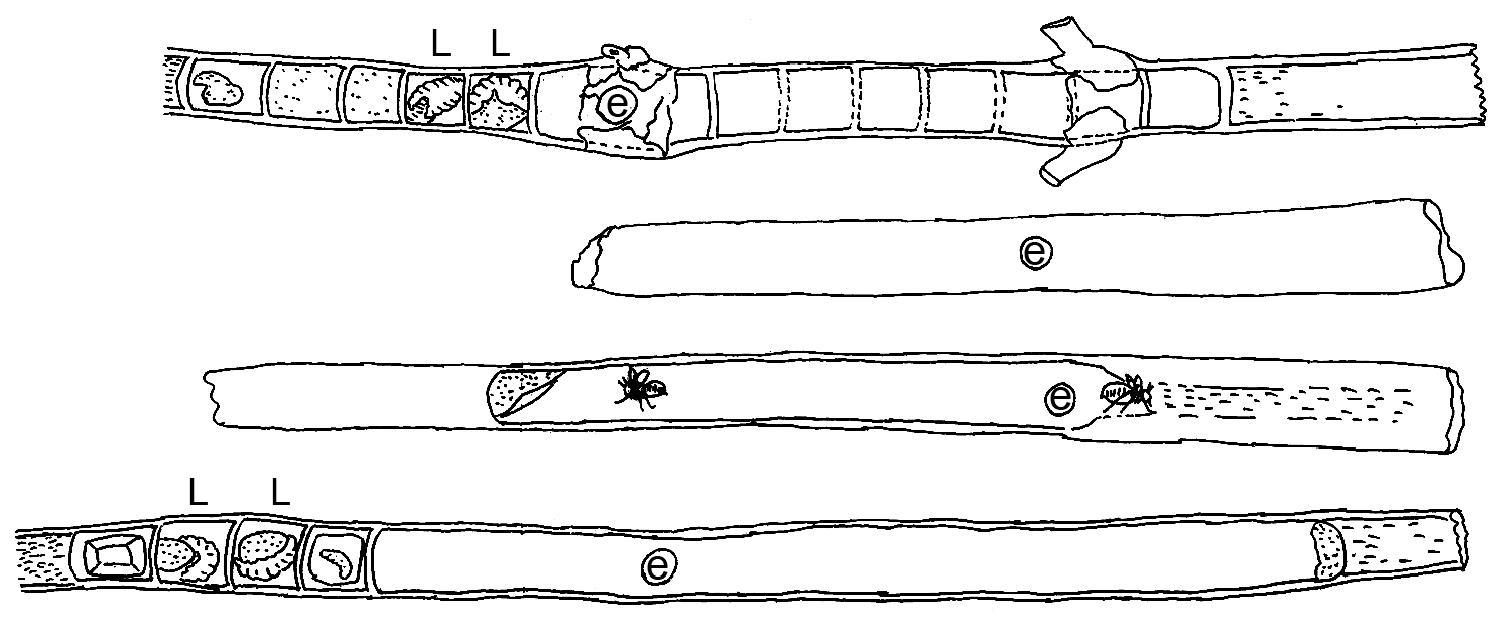
The biology of Xylocopa sulcatipes has been the focus of several extensive ecological and behavioral studies, principally in Israel (e.g., Eisikowitch 1986; Gerling et al. 1983, 1989; Hefetz 1983; Kronenberg and Hefetz 1984; Stark 1989, 1992a, b; Stark et al. 1990; Surholt et al. 1990; Velthuis 1987; Velthuis and Gerling 1980; Velthuis et al. 1984; Willmer 1988). Our observations do not differ from those of the previous studies except that we have focused more on the architecture of the nests rather than the particular ecology of the species, which is already well characterized. The species is bivoltine and foraged from March through November. The area around Amariah, where our observations were made, is a typical central Saudi Arabian desert environment. Vegetation is thinly scattered and comprised mostly native plants, including several promising foraging flowers and nesting sites throughout the season. Among these, Calotropis procera was found to be the most commonly used for nesting and provisioning resources. The pithy and rather straight stems of suitable diameter of Calotropis procera make them ideal for nest construction (e.g., Figs 23–27). Secondarily, Phoenix dactylifera was used as a nesting substrate (with five such nests collected). Detail measurements of the nests observed are provided in table 1. Given the different overall physical structure of these substrates it is not surprising that nests in Calotropis procera had a single, linear nest tube extending to each side of the entrance (Figs 23–27), while those in Phoenix dactylifera consisted of a more gallery-like structure, similar in this regard to the variation observed for Xylocopa (Stenoxylocopa) artifex Smith (Silveira 2002). In all of the nests the pollen masses were compact, well kneaded, and mixed with sufficient nectar to leave them moist (Fig. 26). Where observed, individual pollen loaves were fully consumed and the larvae defecated pellet-like feces which were placed to its back or at the bottom of the cell. As observed elsewhere for this species, cells were arranged linearly, but never with cells closer than 1–1.5 times an individual cell length from the nest entrance. Also similar to observations made elsewhere on this species (e.g., Gerling et al. 1989), some nests were found to comprise several newly emerged females along with an older female, all of whom participated in foraging but apparently built their own cells, although further observations are needed to clarify this point.
Although Xylocopa sulcatipes, like other Xylocopa, is polylectic, females were observed foraging mostly from Calotropis procera and it was there that males were seen to approach and grab females for mating. In addition to foraging at Calotropis procera, females were observed visiting Reseda alba L. (Resedaceae) and radish [Raphanus sativus L. (Brassicaceae)]. Given that species of Xylocopa may be useful for agricultural pollination (Keasar 2010) it may be beneficial for standing crops in central Saudi Arabia to be surrounded by suitable native vegetation including Calotropis procera, thereby providing ample nesting sites to encourage the establishment of sustainable and large populations of these bees. No associated organisms were found among the nests observed.
Photographs of nests of Xylocopa (Ctenoxylocopa) sulcatipes Maa in stems of Calotropis procera (Aiton) in central Saudi Arabia. 23 Nest entrance in stem of Calotropis procera in the wild 24 Opened nest with series of larvae in individual cells 25 Opened nest with pupae 26 Individual pollen mass with egg situated on top.
Photographs of nests of Xylocopa (Ctenoxylocopa) sulcatipes Maa in stems of Calotropis procera (Aiton) in central Saudi Arabia. 23 Nest entrance in stem of Calotropis procera in the wild 24 Opened nest with series of larvae in individual cells 25 Opened nest with pupae 26 Individual pollen mass with egg situated on top.
Diagrams of representative nests of Xylocopa (Ctenoxylocopa) sulcatipes Maa in stems of Calotropis procera (Aiton) in central Saudi Arabia. L = larva; e = nest entrance. Line illustrations by M.A. Hannan.
Diagrams of representative nests of Xylocopa (Ctenoxylocopa) sulcatipes Maa in stems of Calotropis procera (Aiton) in central Saudi Arabia. L = larva; e = nest entrance. Line illustrations by M.A. Hannan.
This work was supported by King Saud University, Deanship of Scientific Research, College of Food and Agriculture Sciences Research Center. We are thankful to Naser Al-Ghoson and Hassan Balhareth who helped us during the course of this study, to Prof. Charles D. Michener for encouragement, to Dr. Ismael A. Hinojosa-Díaz for assistance with photomicrography, support for which was provided by the Engel Illustration Fund of the University of Kansas College of Liberal Arts and Sciences, and to two anonymous reviewers for their comments on the manuscript. Lastly, we are grateful to the owner and employees of the agricultural farm Mazra’ah Al-Gasim who generously permitted us to work on their property. This is a contribution of the Division of Entomology, University of Kansas Natural History Museum.